
WAKIX Pitolisant 17.8MG TAB 30
WAKIX 17.8MG TAB 30 BY HARMONY BIOSCIENCE Pitolisant hydrochloride tablet, film coated Item No.:RX72028-178-03 NDC No. 72028-178-03 72028-0178-03 72028017803 7202817803 UPC No.:3-72028-17803-
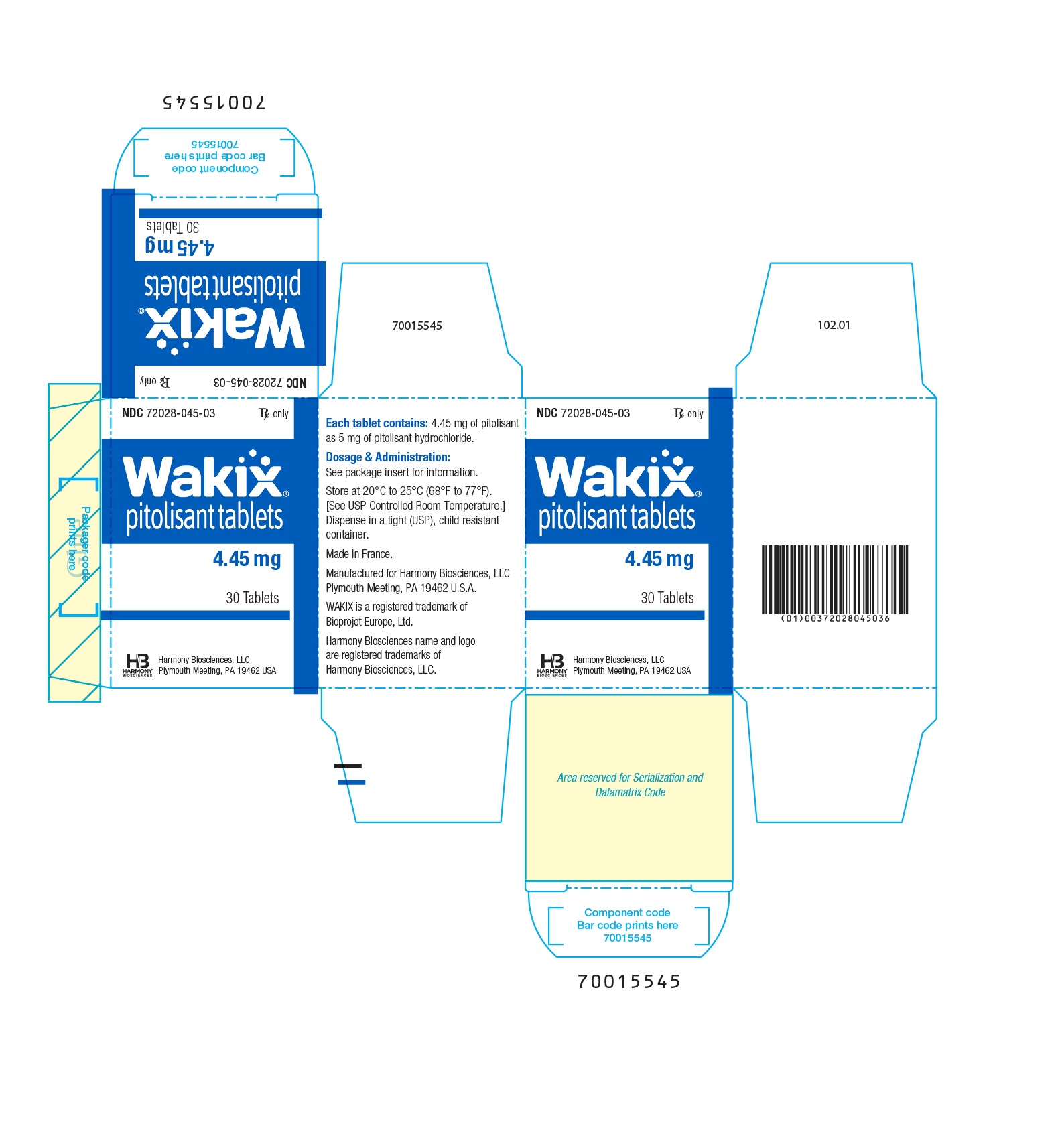
Pitolisant 4.5MG TAB 30
WAKIX 4.5MG TAB 30 BY HARMONY BIOSCIENCE Pitolisant hydrochloride tablet, film coated Item No.:RX72028-045-03 NDC No.72028-0045-03 72028-045-03 72028004503 7202804503 UPC No.:3-72028-04503-6 372028

Buy More Save More!
Want to do Research on this Med or need a large quantity? Email Details with quantity required to:[email protected]

AmericanPharmaWholesale.com
Visit AmericanPharmaWholesale.com for over 100,000 items of Health & Beauty at Retail@Wholesale prices.

Erwinaze 10,000 IU 5Vials/Pack
Erwinaze 10,000 IU 5Vials/Pack B

Erwinaze 10,000 IU 5Vials/Pack
Erwinaze 10,000 IU 5Vials/Pack B

WAKIX Pitolisant 17.8MG TAB 30
WAKIX 17.8MG TAB 30 BY HARMONY BIOSCIENCE Pitolisant hydrochloride tablet, film coated Item No.:RX72028-178-03 NDC No. 72028-178-03 72028-0178-03 72028017803 7202817803 UPC No.:3-72028-17803-

Buy More Save More!
Want to do Research on this Med or need a large quantity? Email Details with quantity required to:[email protected]

SUNOSI 75 MG TAB By Jazz Ph
SUNOSI 75 MG TAB 30 By Jazz Pharma-Solriamfetol HCl ORAL TABLET Item No.:DEA885677 885677 NDC No.68727-0350-01 68727-350-01 6872735001 68727035001 UPC No.:1-03687-2735001-1 103687-27350011 10368727

SUNOSI 75 MG TAB By Jazz Ph
SUNOSI 75 MG TAB 30 By Jazz Pharma-Solriamfetol HCl ORAL TABLET Item No.:DEA885677 885677 NDC No.68727-0350-01 68727-350-01 6872735001 68727035001 UPC No.:1-03687-2735001-1 103687-27350011 10368727

Pitolisant 4.5MG TAB 30
WAKIX 4.5MG TAB 30 BY HARMONY BIOSCIENCE Pitolisant hydrochloride tablet, film coated Item No.:RX72028-045-03 NDC No.72028-0045-03 72028-045-03 72028004503 7202804503 UPC No.:3-72028-04503-6 372028