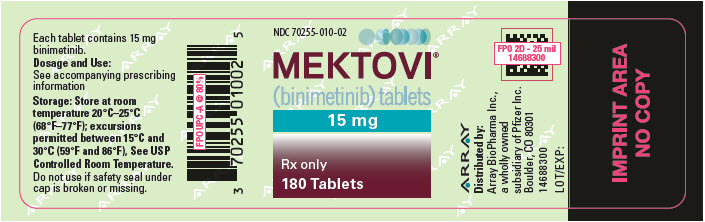
RX ITEM-Mektovi 15Mg (2700 Mg) Tablets 1
Generic Name:
BINIMETINIB
Description:
MEKTOVI, 15MG, (2700 MG), TABLETS, 1X180 EA
Alt Item #:
5436688
NDC:
70255-0010-02
UPC:
370255-010025
Contract:
Contract Alias:
Strength:
15MG
Form

Mektovi 15Mg (2700 Mg) Tablets 1X180 Ea
BRAND: MEKTOVI NDC: RX70255-0010-02 ,RX70255001002 UPC: 370255-010025,370255010025 Generic Name:
BINIMETINIB
Description:
MEKTOVI, 15MG, (2700 MG), TABLETS, 1X180 EA
Alt Item #:
5436688
NDC:

American Pharma Wholesale
Only Lic.-Physician,Pharmacy,Dentist,Drug Mfg,Dist.,Gov,Hospital,Lic.Lab,Naturalist,Naturopath,NP,Optometrist,Pharmacist,PA,Physical Therapist,Podiatrist,Research Co.,Uni.,VA,Vet & Wholesalers in scop

Buy More Save More!
Want to do Research on this Med or need a large quantity? Email Details with quantity required to:[email protected]

AmericanPharmaWholesale.com
Visit AmericanPharmaWholesale.com for over 100,000 items of Health & Beauty at Retail@Wholesale prices.