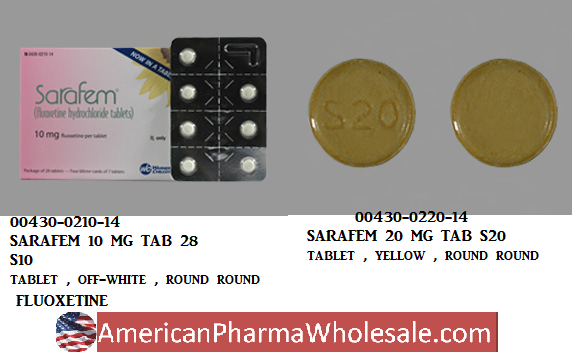
RX ITEM-Sarafem 20Mg Tab 28 By Actavis P
NDC 00430-0220-14 UPC/GTIN No.3-04300-22014-5 Mfg.Part No.22014
Sarafem 20Mg Tab 28 By Actavis Pharma
BRAND: SARAFEM NDC: 00430-0220-14,430022014 UPC: 3-04300-22014-5,304300220145

Click above for FLUOXETINE
Only Lic.-Physician,Pharmacy,Dentist,Drug Mfg,Dist.,Gov,Hospital,Lic.Lab,Naturalist,Naturopath,NP,Optometrist,Pharmacist,PA,Physical Therapist,Podiatrist,Research Co.,Uni.,VA,Vet & Wholesalers in scop

Buy More Save More!
Want to do Research on this Med or need a large quantity? Email Details with quantity required to:[email protected]

AmericanPharmaWholesale.com
Visit AmericanPharmaWholesale.com for over 100,000 items of Health & Beauty at Retail@Wholesale prices.